Disappearing Ink
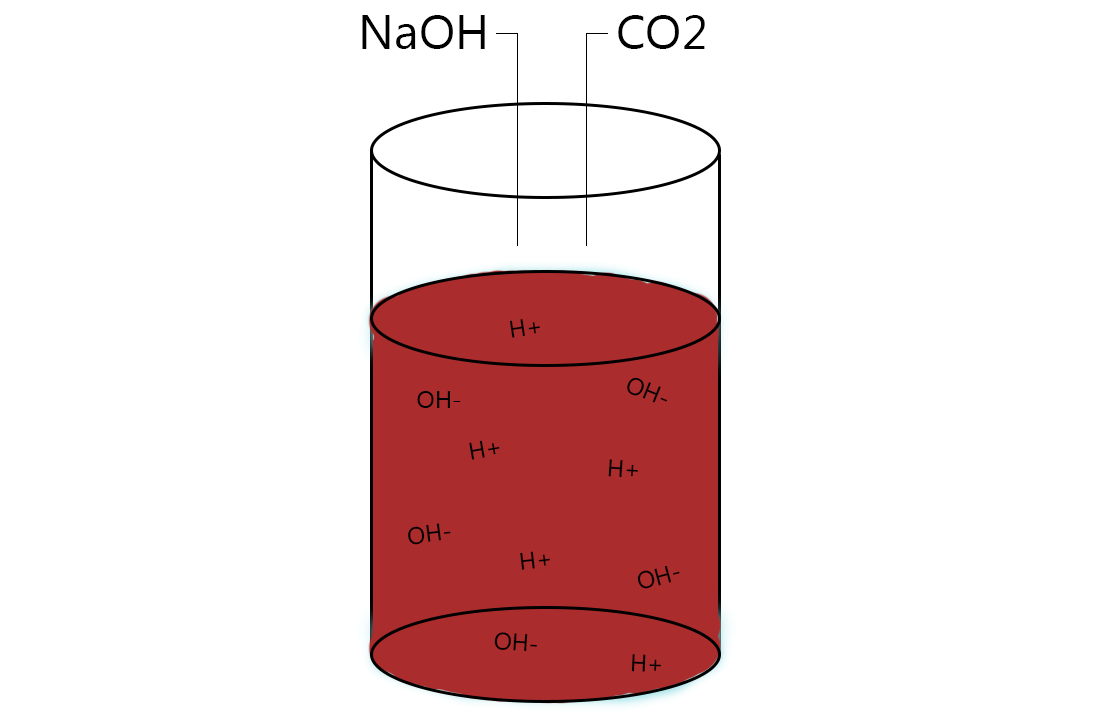
In class, we made invisible ink and learned about how it works. We put 0.1g of phenolphthalein ( a white powder) into a 125 mL erlenmeyer flask. Next, we added 10 mL of ethanol (lab alcohol) and stirred until it dissolved. Then, we added 100 mL of DI H2O (distilled water) and finally, put in 3m (dropwise, about 7 drops) of NaOH (sodium hydroxide) until the mixture was dark red.
And how does it turn from dark red to perfectly clear? When the CO2 (carbon dioxide) enters the mixture, it makes it more acidic, which causes the mixture to go invisible. This ink is a pH indicator, because its color changes as the acidity in the mixture increases or decreases. This reaction is sort of similar to what I walked about in the ocean acidification blog post I wrote below, the effects of co2 in the ocean. By simply adding small drops of NaOH,the levels of co2 in the mixture increase.
And how does it turn from dark red to perfectly clear? When the CO2 (carbon dioxide) enters the mixture, it makes it more acidic, which causes the mixture to go invisible. This ink is a pH indicator, because its color changes as the acidity in the mixture increases or decreases. This reaction is sort of similar to what I walked about in the ocean acidification blog post I wrote below, the effects of co2 in the ocean. By simply adding small drops of NaOH,the levels of co2 in the mixture increase.
Ocean Acidification
Recently, we learned a little bit about ocean acidification, which is causing the ocean levels to rise. A bit of backstory; When the industrial revolution started, the pH level in the ocean lept from 8.25 to 8.14, which doesn't seem like a lot, but it's actually a 30 percent increase (The pH scale goes from 0 to 14, 0 being very acidic, 14 being basic). What's happening is because of pollution, from factories, cars, you name it, a lot of CO2 (Carbon Dioxide) is being released into the air. Then, that CO2 mixes with the water molecules, becomes carbonic acid, and releases a charged hydrogen atom. All of the hydrogen atoms are now free in the water, increasing the pH levels in the ocean. This may just sound like a change of numbers and nothing too serious, but it's not good news for the ocean. This change in pH affects the ocean life, such as coral reefs, mollusks, crabs, etc.
Phases of Matter (Dry Ice)
In class, we were allowed to play with dry ice (keeping safety in mind, of course). Dry ice is interesting, because it's a solid that is colder than ice, but when exposed to room temperature, it starts turning into its former gaseous state (carbon dioxide). This has to do with phase changes.
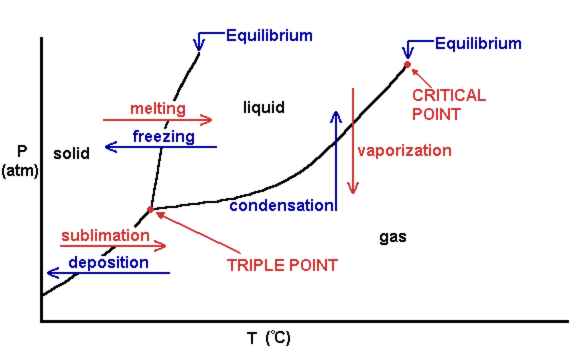
This is a phase change diagram (the y axis is "Pressure" and the x axis is "Temperature"). Andrew taught us that this diagram shows us at which conditions that different phases occur, such as melting, freezing, gas, solid, liquid, etc. occur. Pressure and temperature are the two factors that determine which state a certain substance is at.
Properties of Matter (Floating Candles)
Yesterday in class, we did an experiment about density that made a trashbag fly, using candles and thin strips of balsa wood. Kind of like a hot air balloon. It took a little bit to actually start flying, because the bag needed to fill/heat up. This experiment worked because all of the heated molecules were bouncing against the inside of the bag, which inflates it. In the equation for density, density = mass over volume, and either less mass or more volume = less density. The bag became lighter/less dense than the air around it and began to float. Since we used a big bag and light materials for the base, If it were heavier, or the bag was smaller, it wouldn't have worked.
Properties of Matter II (Making Rocks)
After we first learned about the phases of matter, we learned more specifically about the Earth, as most know (unless you're living under a rock), has four different layers, which are the inner core, outer core, mantle, and crust. And also, all around each of these layers that make up the Earth, is a magnetic field. This magnetic field deflects solar wind that would strip away our ozone layer, which protects us from ultraviolet radiation. Each of these layers consist of different things. The inner core is made of molten nickle. The outer core is made out of molted nickle as well, along with iron. The mantle is made out magma, which is molten rock. The crust is a solid version of the mantle.
Our lab was focused on what happens in the mantle. The mantle is made of 97% silicates, which form the rocks that make up the crust. Oxygen and silicon mix together with metals that come from below for this to happen. We got to sort of recreate this by putting silicates, chemicals, and distilled water into a glass tube. Little crystals or rocks grew upwards from this mixture, and though it was a little bit of a slow process, it was super cool to watch.
Our lab was focused on what happens in the mantle. The mantle is made of 97% silicates, which form the rocks that make up the crust. Oxygen and silicon mix together with metals that come from below for this to happen. We got to sort of recreate this by putting silicates, chemicals, and distilled water into a glass tube. Little crystals or rocks grew upwards from this mixture, and though it was a little bit of a slow process, it was super cool to watch.
What Is Science?
Science is basically trying to understand nature in an unnatural way; through equations and man made machines and making unnatural combinations and stuff like that.
Update:
In order to write a meaningful update, I watched this video.
Update:
In order to write a meaningful update, I watched this video.
This video was about the very basics of science, more opinions and clarifications than science facts, actually, nothing about cells and electricity and stuff like that. So, to my surprise, I found myself agreeing with pretty much everything Neil deGrasse Tyson said. He was talking about how he believes that people should keep themselves informed, not live life purposely under a rock and know the basic things about our world, and that just because we don't understand something doesn't mean we should be scared of it. Also, he brought up an interesting point how scientists are often portrayed as the bad guys, greedy, or just plain crazy, when often times, they can be the ones with the conscience. All around, this was actually a pretty interesting video to watch, even though as I had proved before in my first statement, I am not a huge fan of science. Looking at the cool experiments, sure, but trying to understand it, with all of the equations and stuff, just doesn't particularly interest me. Though I still am not a fan of all that, I am now more open minded about that other side of science, the understanding part. Hearing a scientist explain the more "human" side really made science more appealing to me, and made me less angry about the idea of it. Most of this video spoke to me, but there was something that I couldn't wrap my head around was how he thought that E=MC2 is beautiful. As I stated before, I am not a fan of equations or that side of science, so I just don't really understand, but it was kind of interesting to hear him explaining it. It was a point of view that I hadn't really heard before. After watching this video, my answer to "What is Science?" is that it's simply just gaining more knowledge about our Earth, and ourselves as well in the process. If we pursue science further and look more into our Earth, we may discover things that can help us and our Earth, but on the opposite end, as it has been proved already, we can discover things that will harm it and us, but for the sake of discovery and knowledge, science is a risk I think we should be willing to take.
Hydrogen Balloon
In class, we did a mini experiment, where we put citric acid and magnesium in a tube and sealed it in with a balloon. The chemical reaction filled the balloon with hydrogen and made it super light and floaty, but that wasn't the coolest part of this experiment. When we held the balloon over the flame, it combusted outwards and popped loudly. What I think happened was the hydrogen atoms combined with the oxygen atoms and cause a mini explosion. The popping sound was just from the balloon itself popping, and not anything special happening, obviously, it's just as if you filled a balloon with oxygen or helium and popped it.
Update:
The science behind this experiment was that the elements were combining, as I had said, but in class we learned that it was a little more complicated than just combining. Elements coming together is all about electrons. Electrons are positioned in shells. The first shell contains a max of 2 , the next has a max of 8, then the next 18, and so on, but the last shell has a max of 8. All elements have different amounts of shells and electrons on those shells. These electrons determine the reactivity of the element,
Update:
The science behind this experiment was that the elements were combining, as I had said, but in class we learned that it was a little more complicated than just combining. Elements coming together is all about electrons. Electrons are positioned in shells. The first shell contains a max of 2 , the next has a max of 8, then the next 18, and so on, but the last shell has a max of 8. All elements have different amounts of shells and electrons on those shells. These electrons determine the reactivity of the element,
Combustion Article
To summarize what I got from this article as a whole, combustion is a chemical reaction that releases heat and is very delicate and important to our everyday life, though it causes a lot of problems for our environment, and to this day is still under a lot of different types of testing. What stood out to me about this read was that more things than I thought in real life involved combustion, I always thought of it as a crazy science experiment or a bad car accident ending in an explosion or something, but it turns out to be what produces my electricity, heats my house, among a few other things. It's also something that isn't yet fully controllable, which is interesting.